 
A sample of solid KHP that is weighed, determined to have a mass of 0.7396 +0.0001 g, and dissolved in - 30 mL of deionized water is titrated with the sodium hydroxide solution in a 100-ml burette, (a) If the initial volume of the dispensed sodium hydroxide solution from the burette is 26.5 + 0.1 mL and the final volume is 52.4 +0.1 mL, calculate the concentration and its absolute uncertainty of the sodium hydroxide solution Molarity M (b) Calculate the mass and its absolute uncertainty in grams of sodium hydroxide used to prepare the solution. 2 The molar mass of Hydrogen is 1.008 g/mol. 
Be sure to use borosilicate glass (e.g., Pyrex) and consider immersing the container in a bucket of ice to keep the heat down. Add sodium hydroxide to water do not add water to solid sodium hydroxide. 1 The molar mass of Oxygen is 15.999 g/mol. Stir the sodium hydroxide, a little at a time, into a large volume of water and then dilute the solution to make one liter. 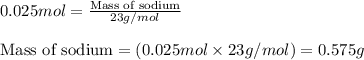
Sodium hydroxide (NaOH, molar mass = 39.997 g/mol, assume to be exact with no uncertainty) solutions are standardized with potassium hydrogen phthalate (KHPh, molar mass=204.221 g/mol, assume to be exact with no uncertainty) by titration procedures involving the following reaction (20 points) KHPh(aq) + NaOH(aq) → H2O(l) + Naph(aq) A 350.00 mL (assume this volume is exact with no uncertainty) sodium hydroxide solution is standardized by this approach. The molar mass of Sodium is 22.990 g/mol. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
